This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
|
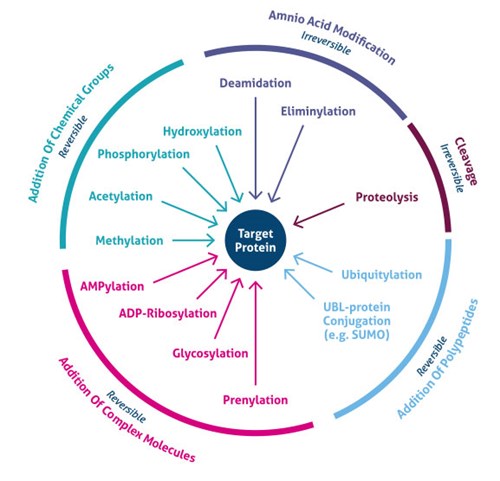
As of recently it's been discovered that the proteome is much more complicated than the genome. Well 20,000 to 25,000 genes are known, the total amount of proteins is estimated at over 1 million. This is due to mechanisms such as splicing, alternative promoters, and other slight alterations made to the transcription and translation process. This increase in complexity of the proteome is also due in part to post-translational modifications (PTM) [1]. What are Post-Translational Modifications? [1] Post-translational modifications are the addition of functional groups to already translated proteins that results in a wide range of properties. They can regulate such characteristics of proteins such as localization, activity, and interactions with other molecules. Often times the addition of PTMs is regulated by enzymatic activity and occur only at specific amino acids. Such modifications can occur at any point in the life span of a protein. Sometimes proteins are modified right after being made while others occur as a result of stimuli later on. Some common PTMs include phosphorylation, acetylation, and methylation. |
|
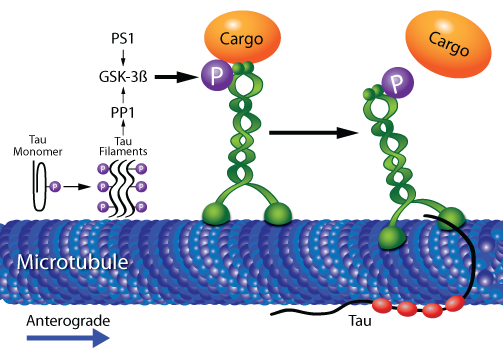
Phosphorylation of KIF5A [2] PTM of the cargo binding region of both the heavy and light chain of Kinesin proteins has been shown to regulate transport. The Kinesin motor domain is also another region of the protein that is typically modified by phosphate groups. When phosphorylated, research has suggested that kinesin favors transport towards the negative end of the microtubule much like how dynein transports cargo. |
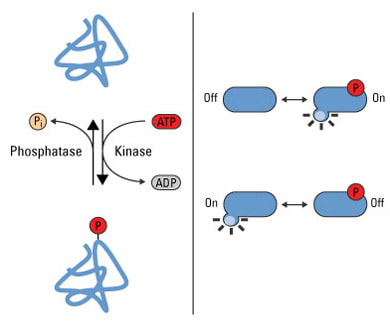
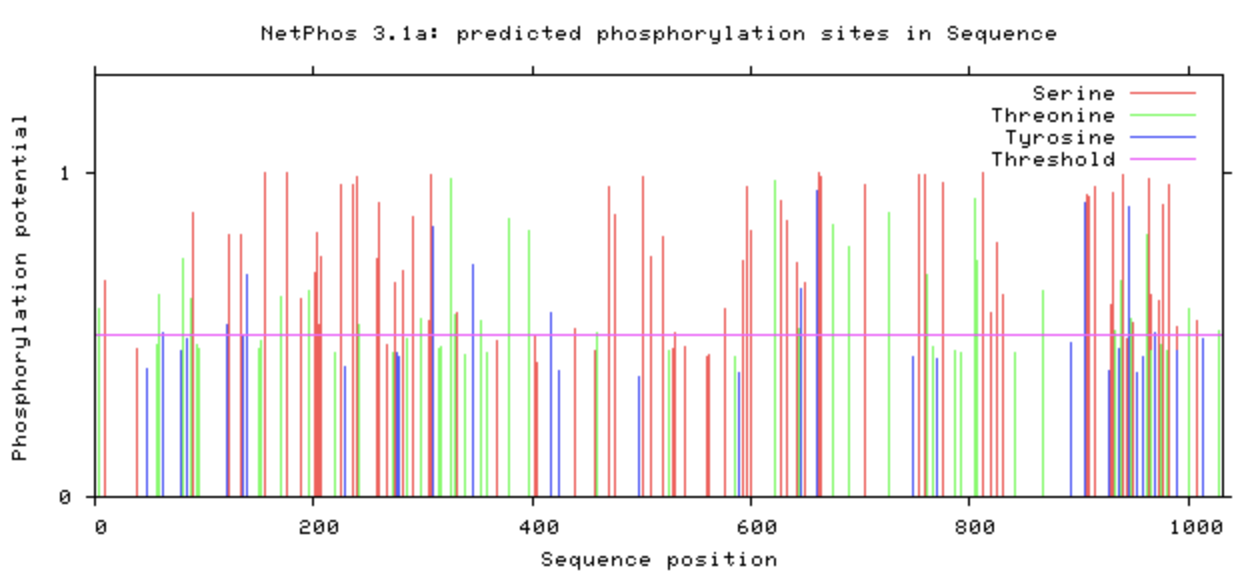
What is Phosphorylation? [1] Phosphorylation is the addition of a phosphate group to a protein. The addition of phosphate groups is reversible and often functions as a regulator of activity for a protein attaching typically at Serine, Tyrosine, and Threonine. This type of PTM is usually found in processes such as the cell cycle, apoptosis, and signal transduction pathways. NetPhos Phosphorylation Sites The NetPhos output for KIF5A gives the predicted sites of phosphorylation on the protein. Any lines that fall above the pink threshold line show that there is a high confidence for that predicted site. According to the database, KIF5A is phosphorylated all along the protein with a high concentration near the C-terminus. |
Conclusion
As previous research has suggested concerning KIF5A, the C-terminus tends to be phosphorylated in order to regulate cargo transport in cells. This information is seems to coincide with the predicted sites that NetPhos provided on their database. This information is useful in understanding how KIF5A functions normally in a healthy cell.
References:
[1] Overview of Post-Translational Modifications. (n.d.) Retrieved from https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
[2] Musumeci, O., Bassi, M.T., Mazzeo, A. et al. Neurol Sci (2011) 32: 665. https://doi.org/10.1007/s10072-010-0445-8
[1] Overview of Post-Translational Modifications. (n.d.) Retrieved from https://www.thermofisher.com/us/en/home/life-science/protein-biology/protein-biology-learning-center/protein-biology-resource-library/pierce-protein-methods/overview-post-translational-modification.html
[2] Musumeci, O., Bassi, M.T., Mazzeo, A. et al. Neurol Sci (2011) 32: 665. https://doi.org/10.1007/s10072-010-0445-8