This web page was produced as an assignment for Genetics 564, an undergraduate capstone course at UW-Madison.
What are Protein Domains? [1]
Domains are distinct functional units found within a protein that are often times conserved across species. Proteins can have multiple domains that help play a role in interactions in a biological system. These subunits tend to function separate from the rest of the amino acid chain, typically existing in a variety of proteins that have different overall functions. Protein domains are a good indicator of evolution as a result of how conserved the regions are when compared to other species.
KIF5A Protein Domains [2]
|
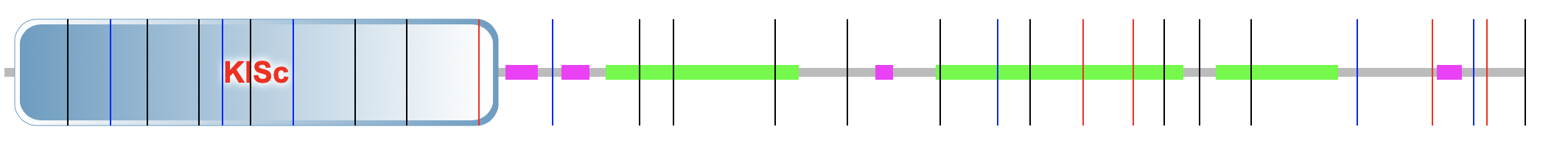
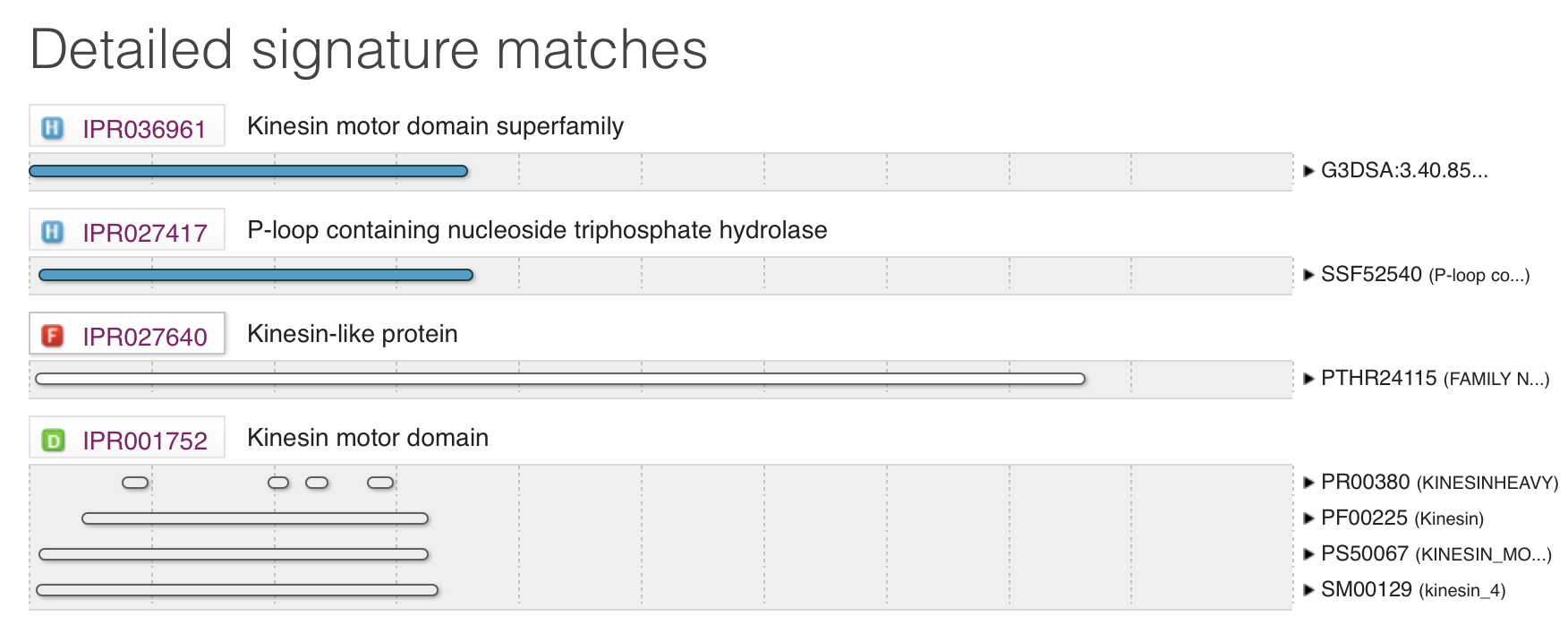
KIF5A was analyzed using Pfam, SMART, and InterPro in order to find different domains on the protein. All three programs were utilized to compare and make sure all domains and significant regions were identified. Only one domain on KIF5A was found swell as a few low complexity and coiled coil regions. The only domain noted to be on the protein is KISc: a Kinesin motor domain. The figures on the right depict the results from SMART and InterPro.
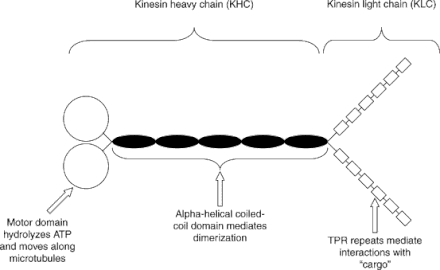
What is Kinesin? Kinesin is a microtubule associated force-producing protein that plays a role in in organelle transport. The motor activity of the Kinesin is typically directed towards the plus end of microtubules. This complex is composed of two heavy chains and two light chains. The heavy chain in particular is typically composed of three structural domains: the KISc domain, a coiled coil region that mediates chain dimerization, and a C-terminal domain that interacts with other proteins vesicles and organelles. All three programs were unable to identify the C-terminal domain on KIF5A; only the Kinesin Motor was illustrated as the major domain on the protein. |
SMART
InterPro
KISc domain
The Kinesin motor domain is located on the heavy chain of Kinesin proteins. This is a large globular N-terminal domain which is responsible for the motor activity of the Kinesin by hydrolysis of ATP so the domain binds and moves along the microtubule. The domain consists of several motifs including N1 (P-loop), N2 (Switch II), N3 (Switch II), N4 and L2 (KVD finger). It has a mixed eight stranded beta-sheet core with flanking solvent exposed alpha-helices and a small three-stranded antiparallel beta-sheet in the N-terminal region. The homologous Kinesin motor domain superfamily is found in 85215 proteins, so the domain it self is very well conserved across many organisms. The conservation of this domain is logical because transport of organelles and other cell cargo is essential to cell functionality. The domain also helps explain the the biological role that KIF5A plays cells. |
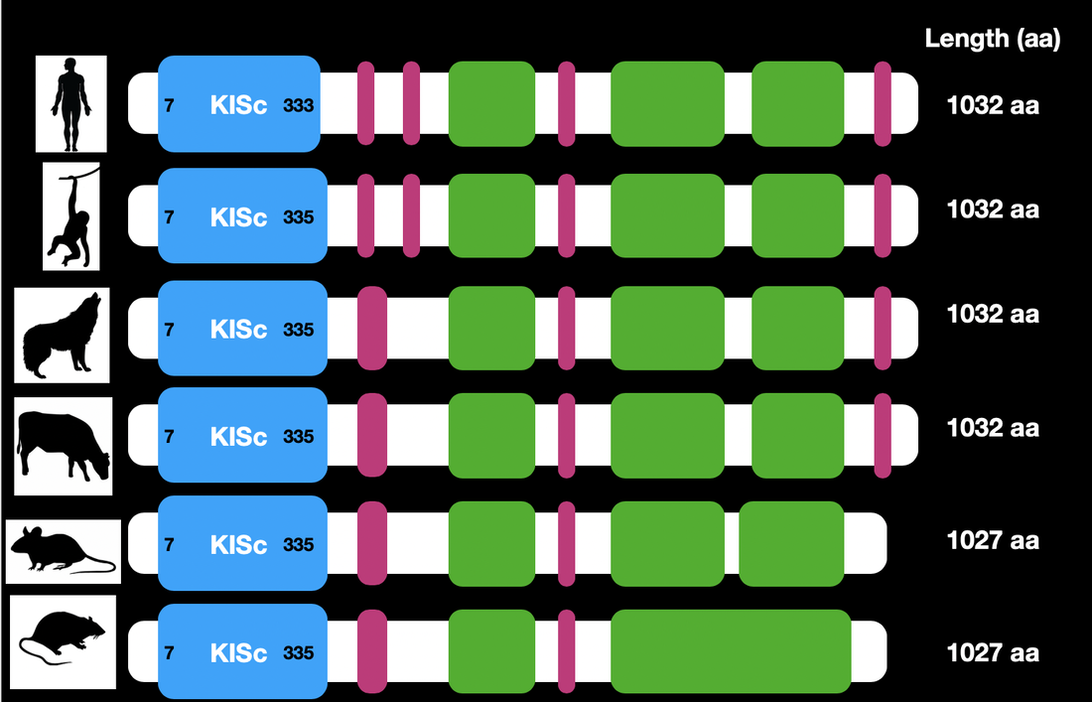
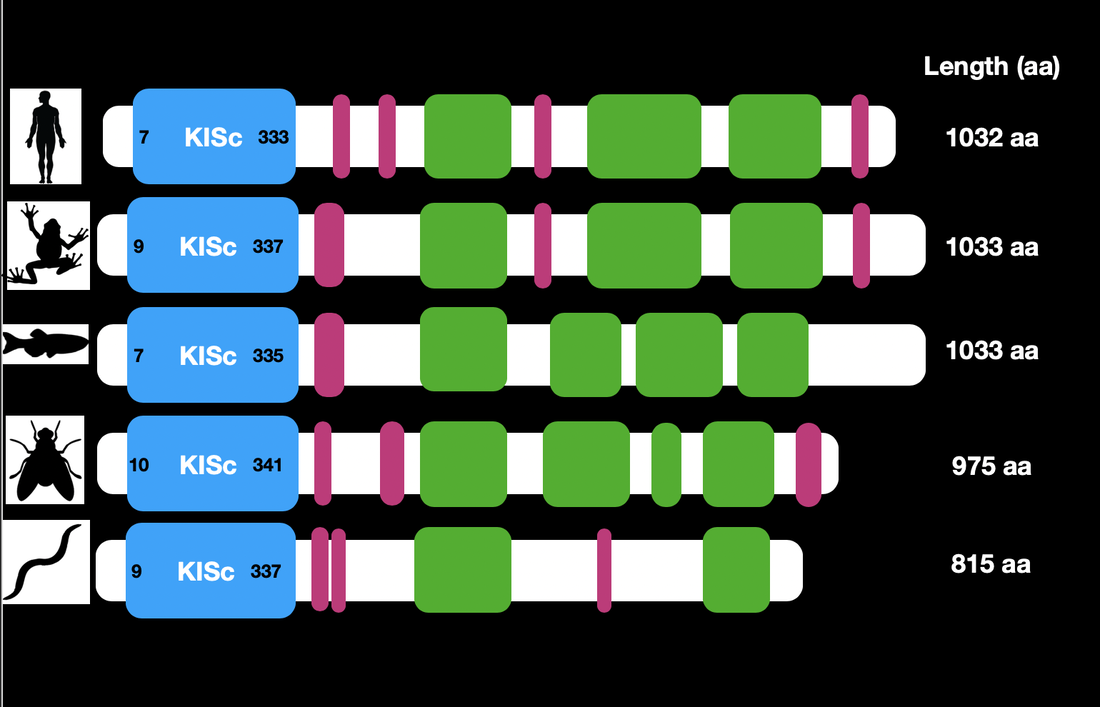
Conservation of KIF5A Domains in Homologs
Conclusion
Domain and certain characteristics of KIF5A were found to be well conserved along several homologs. Notably the Kinesin domain is well-conserved in zebra fish, our model organism. The conservation of the protein amongst all of the homologs suggests that the domains play a key role in the function of the protein and its role in its biological system. This information also helps solidify the relatedness of each organism to one another.
References:
[1] What are Protein Domains. (n.d.) Retrieved from https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
[2] Kinesin Motor Domain. (n.d.) Retrieved from http://www.ebi.ac.uk/interpro/entry/IPR001752
[1] What are Protein Domains. (n.d.) Retrieved from https://www.ebi.ac.uk/training/online/course/introduction-protein-classification-ebi/protein-classification/what-are-protein-domains
[2] Kinesin Motor Domain. (n.d.) Retrieved from http://www.ebi.ac.uk/interpro/entry/IPR001752